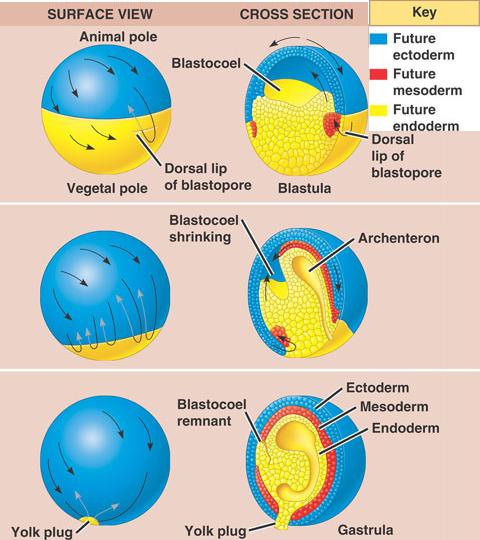
In the limb bud, Wnt5a signaling gradient controls limb elongation by establishing PCP in chondrocytes along the proximal-distal axis through regulating Vangl2 phosphorylation. Wnts dose-dependently induce Vangl2 phosphorylation of serine/threonine residues and Vangl2 activities depend on its levels of phosphorylation. Vangl2, a core component in planar cell polarity, forms Wnt-induced receptor complex with Ror2 to sense Wnt dosages. Here, we show that Wnt signaling gradient provides directional information to a field of cells. However, it is largely unknown whether and how signaling gradients also control cell polarities by acting as global cues. Morphogen gradients can generate different cell types in specific spatial order at distinct threshold concentrations. It is fundamentally important that signaling gradients provide positional information to govern morphogenesis of multicellular organisms. Our results suggest that opposing gradients of Wnt5a and Wnt5b and of their Sfrp inhibitors, together with intercellular signaling via PCP proteins, polarize node cells along the anterior-posterior axis for breaking of left-right symmetry. The absence of planar cell polarity (PCP) core proteins Prickle1 and Prickle2 in individual cells or local forced expression of Wnt5a perturbed polarization of neighboring wild-type cells. Here we show that polarization of node cells is impaired in Wnt5a–/–Wnt5b–/– and Sfrp mutant embryos, and also in the presence of a uniform distribution of Wnt5a or Sfrp1, suggesting that Wnt5 and Sfrp proteins act as instructive signals in this process. Wnt5a and Wnt5b are expressed posteriorly relative to the node, whereas genes for Sfrp inhibitors of Wnt signaling are expressed anteriorly. How node cells become polarized has remained unknown, however. Polarization of node cells along the anterior-posterior axis of mouse embryos is responsible for left-right symmetry breaking. Published by The Company of Biologists Ltd. These findings support a model in which the Shroom3 and PCP pathways interact to control CE and polarized bending of the neural plate and provide a clear illustration of the complex genetic basis of NTDs. Finally, we demonstrate that Shroom3 mutant embryos exhibit defects in planar cell arrangement during neural tube closure, suggesting a role for Shroom3 activity in CE. We observed that multiple components of the Shroom3 pathway are planar polarized along mediolateral cell junctions in the neural plate of E8.5 embryos in a Shroom3 and PCP-dependent manner. We further show that these pathways intersect at Dishevelled, as Shroom3 and Dishevelled 2 co-distribute and form a physical complex in cells. In embryos, simultaneous depletion of Shroom3 and the PCP components Vangl2 or Wnt5a results in an increased liability to NTDs and CE failure. Here we investigate the role of Shroom3 as a direct linker between PCP and actomyosin contractility during mouse neural tube morphogenesis. These complicated processes require the coordinated activities of Rho-Kinase (Rock), to regulate cytoskeletal dynamics and actomyosin contractility, and the Planar Cell Polarity (PCP) pathway, to direct the polarized cellular behaviors that drive convergent extension (CE) movements. Neural tube closure is a critical developmental event that relies on actomyosin contractility to facilitate specific processes such as apical constriction, tissue bending, and directional cell rearrangements. The PCP cue is distinct from neural inducers and has a planar mode of transmission. The dorsal blastopore lip is likely the source of the PCP-instructing signal for the Xenopus neural plate. The Xenopus neural plate progressively acquires PCP in a posterior-to-anterior direction. These observations suggest that neuroectodermal PCP is not instructed by a preexisting molecular gradient, but induced by a signal from the dorsal blastopore lip. The PCP cue did not depend on the orientation of the graft and was distinct from neural inducers. Tissue transplantations indicated that PCP is triggered in the neural plate by a planar cue from the dorsal blastopore lip. By imaging Vangl2 and Prickle3, we show that PCP is progressively acquired in the neural plate and requires a signal from the posterior region of the embryo. Here we investigate the Xenopus neural plate, a tissue that has been previously shown to exhibit PCP. Although the segregation of PCP components to opposite cell borders is believed to play a critical role in this pathway, whether PCP derives from egg polarity or preexistent long-range gradient, or forms in response to a localized cue remains a challenging question. Coordinated polarization of cells in the tissue plane, known as planar cell polarity (PCP), is associated with a signaling pathway critical for the control of morphogenetic processes.
